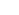우리학과는 KAIST Bio-IT Healthcare Initiative III 사업의 일환으로 11/21일과 12/3일 Inspiring Talk Series를 개최하였습니다.
11/21일 UIUC공현준 교수의 강연은 생체모사 나노 소재를 이용한 의료기기 멸균프로세스에 적용한 연구에 대한 발표와 심층적 토론이 이루어졌으며, 새로운 접근을 통해서 나노소재의 의료기기 응용에 관한 사례연구를 통해 향후 공동연구를 진행할수 있는 가능성을 모색하는 자리가 되었습니다.
12/3일에는 Dr. Rajib Schubert(University of Zurich)를 초청하여 나노툴을 이용한 신경공학 및 뇌신경 맵핑/이미징 기술에 대한 연구에 대한 강연이 진행되었으며, 바이러스를 이용한 세포치료/세포이미징/세포분석 등 다양한 기능에 대해서 알수 있는 계기를 마련하고, 이를 향후 학과의 많은 연구분야와 협업연구로 발전할수 있는 기회로 발전시키기 위한 논의가 이루어졌습니다.
11.21(목) Engineering Active Matters for Biotransport
- Speaker: Prof. Hyunjoon Kong (Department of Chemical and Biomolecular Engineering, Institute for Genomic Biology, UIUC)
- Date and Time : 11AM, Nov. 21 (Thur)
- Place : E16 Chung Moon Soul bldg. room 205(2F)
- Abstract:
Active matters capable of converting chemical energy to mechanical energy present unique features such as self-locomotion and self-manipulation. Active matters can be assembled by controlling the spatial organization of biological cells or chemical reactions of synthetic materials. These matters can discover new physics rules and also have potentials to overcome various technical hurdles in advancing diagnosis and treatments of diseases. We have been working on assembling living matters that reproduce physiological functionality of tissue and synthetic matters that modulate molecular transport across biophysical barriers. The first part of this talk will focus on introducing the role of cross-talks between muscle cells and neurons on recreating neuron-innervated muscle and using it to fabricate biomachinery (Figure 1a). The resulting muscle controls contraction autonomously in response to the neural impulse. The second part of this talk will cover colloidal particles that can remove biofilms formed in confined spaces with their self-locomotion (Figure 1b).
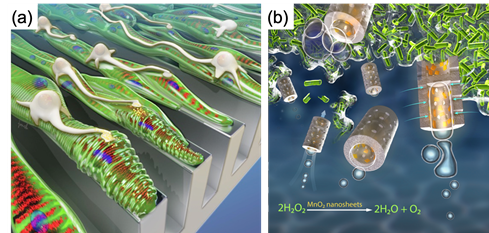
Figure 1. Schematics describing self-locomotive, neuron-innervated muscle (a) and catalytic microbubblers cleaning biofilms (b).
Biography Hyunjoon Kong is a Robert W. Schaffer professor in the Department of Chemical and Biomolecular Engineering, Carle Illinois College of Medicine, and Pathobiology at the University of Illinois at Urbana-Champaign (UIUC). He also holds affiliation with the Department of Bioengineering, Center for Biophysics and Computational Biology, and Neuroscience Program. He received his engineering education from the University of Michigan at Ann Arbor (Ph. D.) and performed post-doctoral research at the University of Michigan and Harvard University. He joined the University of Illinois in 2007. During the academic life, he received the Scientist Development Grant from the American Heart Association, the Career Award from NSF, Center for Advanced Study Fellowship, UIUC Engineering Dean’s Award for Research Excellence (twice), Centennial Scholar, and Promotion Award. He was recently elected to an American Institute of Medical and Biological Engineering (AIMBE) Fellow. Up to dates, he has published 152 papers in various peer-reviewed journals.
12/3(화) Nanotools for neurobiotechnology
- Speaker: Dr. Rajib Schubert, (University of Zurich)
- Date and Time : 11AM, Dec. 3 (Tue)
- Place : E16 Chung Moon Soul bldg. room 205(2F)
- Title: Nanotools for neurobiotechnology
- Abstract :
I will present 3 nanotechnology-based tools I have developed to advance our understanding of complex biological systems such as the brain and cancer at the single cell and multicellular level. The first tool is called ‘virus stamping’, in which viruses are reversibly bound to a delivery vehicle that is brought into physical contact with the target cell on a surface or in tissue, using magnetic forces. Different single cells in the same tissue can be infected with different viruses and an individual cell can be simultaneously infected with different viruses. I use rabies, lenti, herpes and adeno-associated viruses to drive expression of fluorescent markers or a calcium indicator in target cells in cell culture, retina, human brain organoid and the brain of live animals. Virus stamping provides a versatile solution for targeted single-cell infection of diverse cell types, both in vitro and in vivo. The second tool is the atomic force microscope (AFM) in combination optical microscopy to quantify forces of receptor ligand interactions. This combinatorial approach uses a probing tip functionalized with a ligand to image living cells at high-resolution with optical imaging, and simultaneously localize and characterize specific ligand–receptor binding events using the AFM. I will present two applications of this AFM based nanotool: first, with mapping virus receptor interactions in living cells, and second, measuring neuronal cell-cell adhesion receptor – ligand interactions. The last and third tool that I will talk about is the use of novel recombinant binders called DARPins. DARPins (an acronym for designed ankyrin repeat proteins) are genetically engineered antibody mimetic proteins typically exhibiting highly specific and high-affinity target protein binding. I will present two applications DARPins: first, DARPins to engineer viruses for cell type specific infections, and second, as novel optical imaging probes. I will end by giving an overview of how I plan on integrating and further developing the tools presented here to investigate mechanisms of synaptic adhesion proteins in the context of neuronal cell cell adhesions and their role in mediating cellular communication.